By Senior Editor, China Africa News
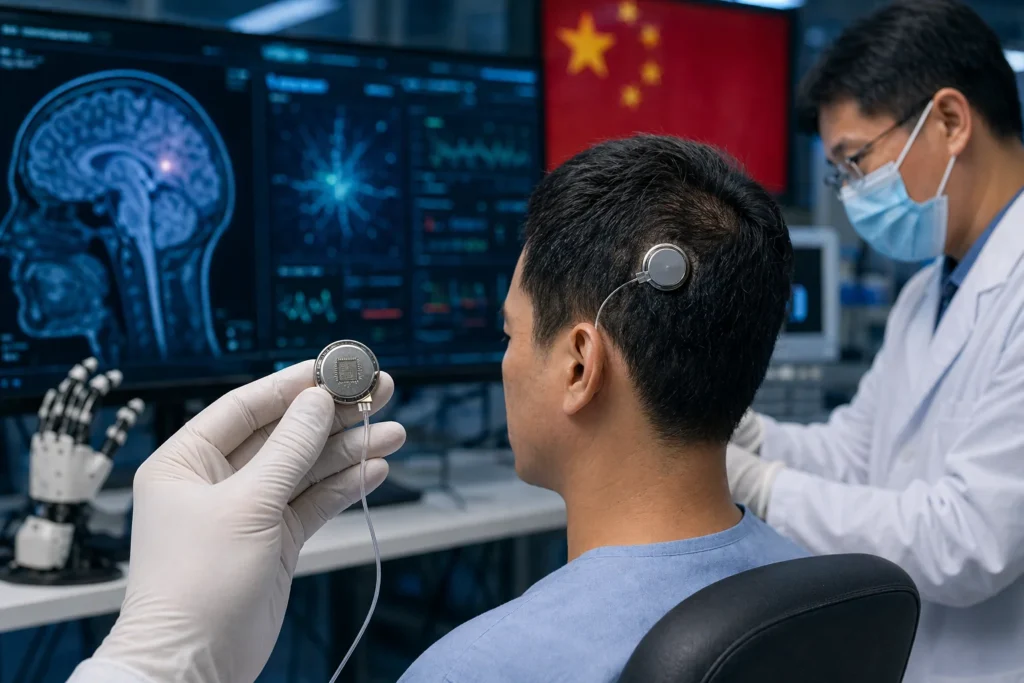
Beijing, April 22, 2026 — China has taken a decisive step into the future of medical technology, approving what is being described as the world’s first implantable brain–computer interface (BCI) for clinical use. The development signals a transition long anticipated by scientists and clinicians alike: the movement of brain–machine communication from experimental settings into practical healthcare.
At the center of this milestone is a device designed to restore lost motor function in patients living with paralysis, particularly those affected by spinal cord injuries. Roughly the size of a coin, the implant is positioned through a minimally invasive procedure outside the brain’s protective membrane. This approach reduces surgical risk while still enabling the device to capture neural signals with sufficient precision.

Once implanted, the system translates brain activity into actionable commands. These signals can then control assistive technologies such as robotic or pneumatic gloves, allowing users to perform everyday tasks grasping objects, lifting a cup, or even writing through intention alone. Early clinical outcomes suggest not only functional improvement but also the possibility of neural adaptation, hinting at deeper rehabilitation potential beyond mechanical assistance.
The approval follows a series of clinical trials involving dozens of patients, all of whom demonstrated measurable gains in hand movement and coordination. In some cases, patients regained a degree of independence that had previously been out of reach, underscoring the transformative implications of the technology.
Beyond its immediate medical benefits, the development marks a broader shift in the global race to harness brain–computer interfaces. Until now, much of the progress in this field has been confined to research laboratories or early-stage experimental trials. Regulatory approval introduces a new phase one defined by real-world application, scalability, and integration into healthcare systems.
The implications extend well beyond paralysis treatment. Researchers are already exploring how similar systems might aid recovery from stroke, support patients with neurodegenerative conditions, or even enhance communication for individuals unable to speak. As the technology matures, it may reshape the boundaries between human cognition and machine capability.
At the same time, the advance raises important questions about ethics, accessibility, and long-term safety. Implantable devices that interact directly with brain signals inevitably prompt discussions about data privacy, consent, and the potential for misuse. Ensuring that such innovations remain focused on therapeutic benefit while addressing these concerns will be critical as adoption expands.
For now, the approval represents a landmark achievement one that demonstrates both the pace of innovation in neurotechnology and its growing readiness for practical use. What was once the domain of science fiction is steadily becoming a clinical reality, offering new possibilities to individuals whose lives have been constrained by physical limitations.